Counterion Distribution around Hydrophilic Molecular Macroanions: The Source of the Attractive Force in Self‐Assembly - Pigga - 2009 - Angewandte Chemie International Edition - Wiley Online Library

Counterion Distribution around Hydrophilic Molecular Macroanions: The Source of the Attractive Force in Self‐Assembly - Pigga - 2009 - Angewandte Chemie International Edition - Wiley Online Library
Spectroscopy, microscopy, diffraction and scattering of archetypal MOFs: formation, metal sites in catalysis and thin films - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00635A

PDF) Optical response of mixed methylammonium lead iodide and formamidinium tin iodide perovskite thin films
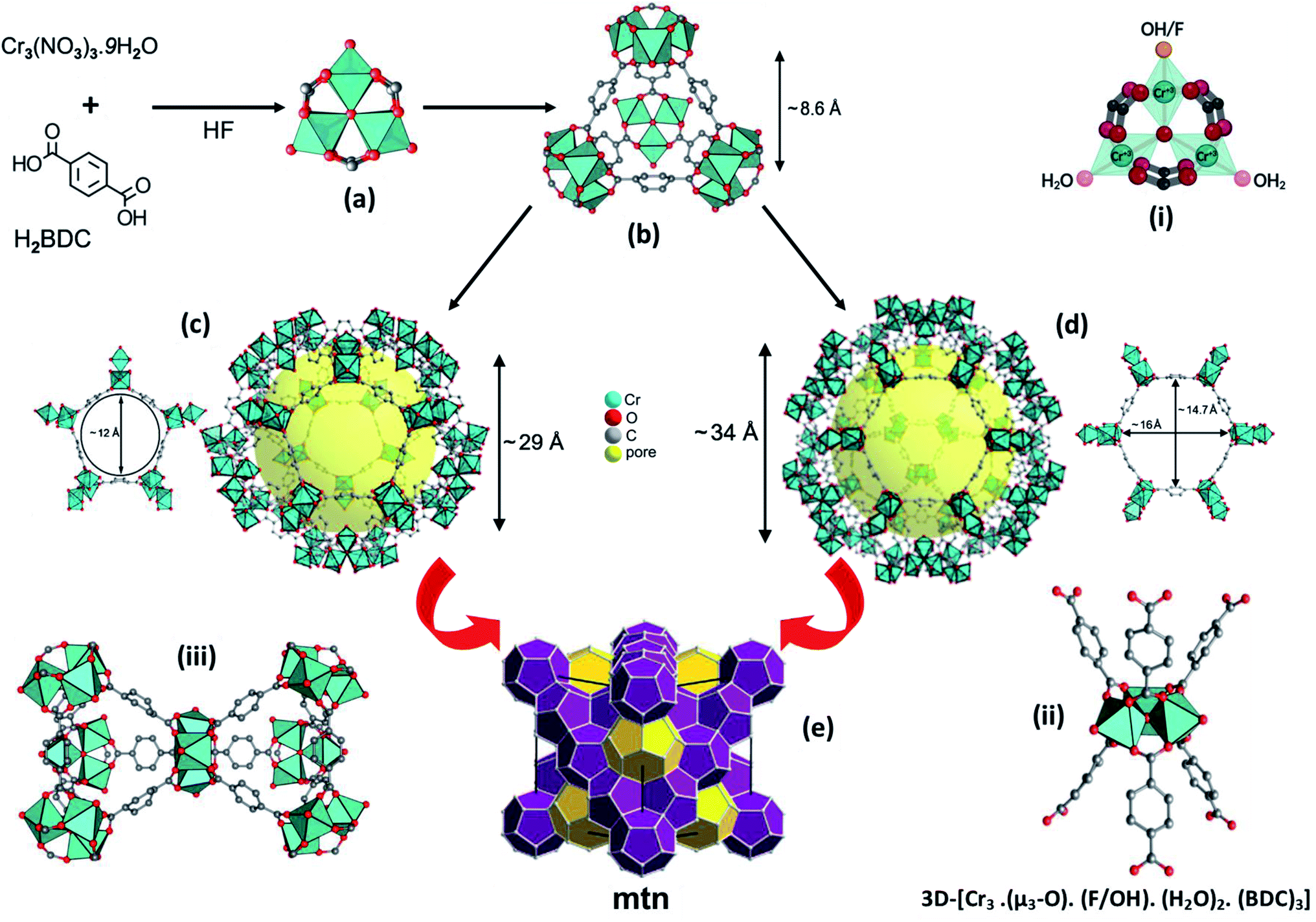
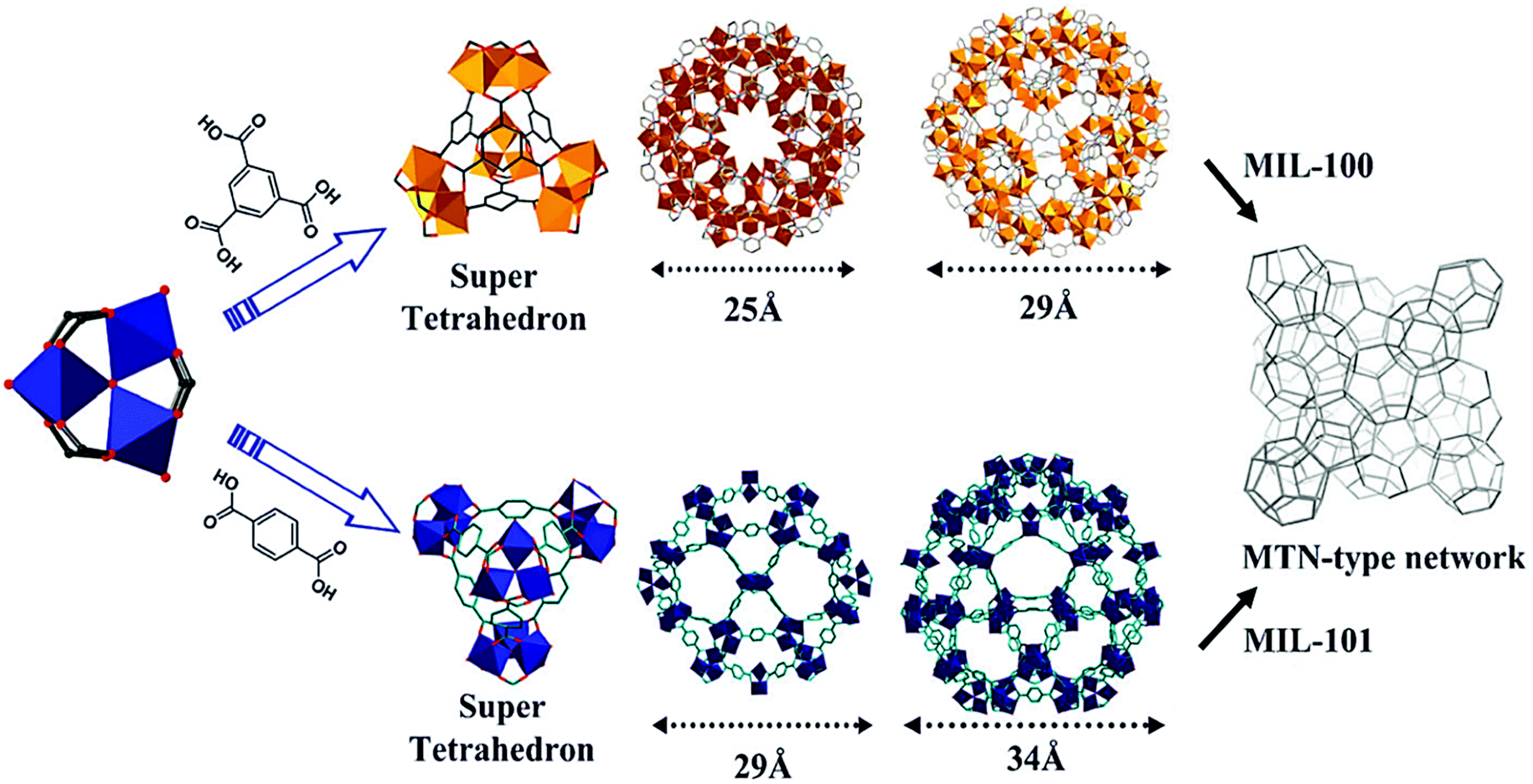
Revisiting the MIL-101 metal–organic framework: design, synthesis, modifications, advances, and recent applications - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/D1TA06238G

Selective Permeability of Uranyl Peroxide Nanocages to Different Alkali Ions: Influences from Surface Pores and Hydration Shells - Gao - 2015 - Chemistry – A European Journal - Wiley Online Library