Inhaled budesonide in the treatment of early COVID-19 (STOIC): a phase 2, open-label, randomised controlled trial - The Lancet Respiratory Medicine

These highlights do not include all the information needed to use budesonide inhalation suspension safely and effectively. See full prescribing information for budesonide inhalation suspension.BUDESONIDE inhalation suspension Initial U.S. Approval: 2000

Teva Launches Two Digital Inhalers in the U.S., AirDuo® Digihaler® (fluticasone propionate and salmeterol) Inhalation Powder and ArmonAir® Digihaler® (fluticasone propionate) Inhalation Powder | Business Wire
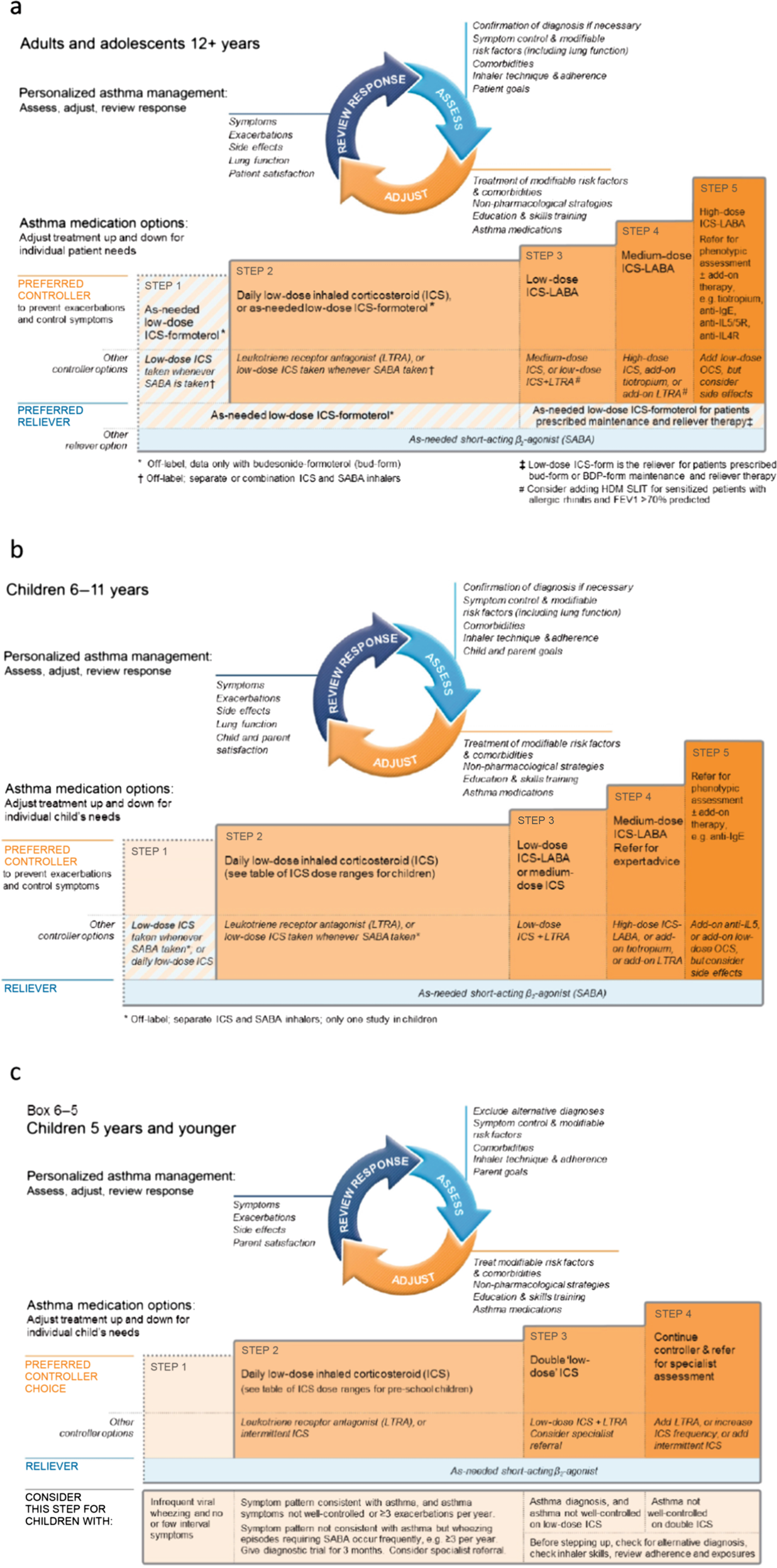
Comparing LAMA with LABA and LTRA as add-on therapies in primary care asthma management | npj Primary Care Respiratory Medicine

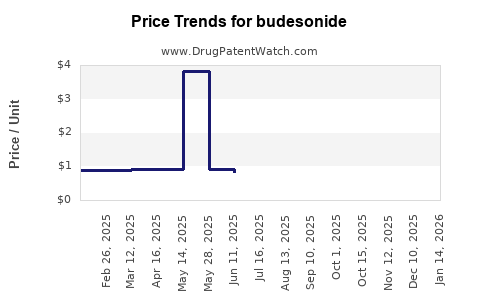
Real-life effectiveness and safety of the inhalation suspension budesonide comparator vs the originator product for the treatment of patients with asthma: a historical cohort study using a US health claims database. -